what would happen to the molality of cyclohexane if some of it evaporated
Lab seven - Determination of the Molar Mass of an Unknown Solid by Freezing Signal Low
Goal and Overview
In the first part of the lab, a series of solutions will be made in order to decide the freezing point depression constant, Chiliad f, for cyclohexane. The freezing points of these solutions, which will comprise known amounts of p-dichlorobenzene dissolved in cyclohexane, will be measured. In the second function of the lab, the freezing point of a cyclohexane solution volition be prepared that contains a known mass of an unknown organic solid. The measured freezing signal change will exist used to summate the molar mass of the unknown solid.
Objectives and Science Skills
-
•
Sympathise and explain colligative backdrop of solutions, including freezing point depression. -
•
Measure the freezing point of a pure solvent and of a solution in that solvent containing an unknown solute. -
•
Calculate the molar mass of the unknown solute and decide its probable identity from a prepare of choices. -
•
Quantify the expected freezing point alter in a solution of known molality. -
•
Quantitatively and qualitatively compare experimental results with theoretical values. -
•
Place and hash out factors or effects that may contribute to the uncertainties in values determined from experimental information.
Suggested Review and External Reading
-
•
Reference information on thermodynamics; information analysis; relevant textbook data
Groundwork
If a liquid is at a higher temperature than its environs, the liquid'south temperature will fall as it gives up heat to the surroundings. When the liquid's temperature reaches its freezing signal, the liquid'southward temperature will not fall as it continues to surrender rut to the surroundings. Instead, the liquid volition brainstorm to freeze. Those parts closest to the cold environment will turn from liquid to solid. This freezing process releases a quantity of rut free energy that is equal in magnitude simply opposite in sign to the heat of fusion, Δ H fusion, of the substance. Δ H fusion is defined every bit the heat captivated by a solid in the process of melting. When the liquid freezes to form the solid stage, it must surrender the same amount of oestrus: Δ H freezing = – Δ H fusion. The temperature of the arrangement (the freezing liquid) remains at the freezing point until all of the liquid has frozen. If yous plot temperature confronting fourth dimension for this process, the bend will resemble that shown. Once the liquid has completely frozen, only then will the temperature start to fall again.
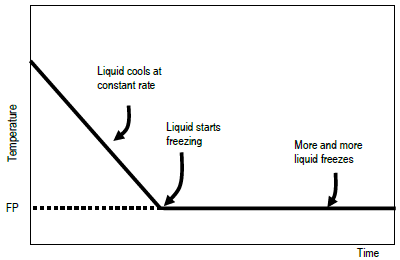
Figure one : Idealized plot of temperature vs. time to make up one's mind freezing
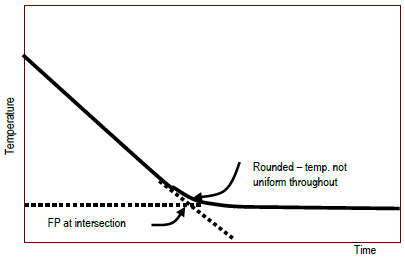
Figure 2 : Typical experimental plot of temperature vs. time to decide point
Typical arcadian and experimental plots are shown. The temperature curve on the experimental plot is rounded, rather than sharp. The system has warmer and colder regions even though it is existence stirred, but the thermometer can merely measure an average temperature. To extract a unique freezing point from the rounded corner on the plot, straight lines are drawn that extend the steeply falling office and the nearly horizontal role of the curve. The experimental freezing point is the temperature at which these two lines intersect. This is shown in Effigy ii. The freezing point of a liquid and the melting point of a solid occur at the same temperature. This temperature is divers equally the point at which the solid and liquid phases coexist in equilibrium with each other under a given pressure level. This is shown in Figure 3.
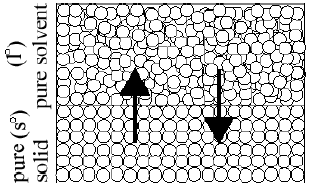
Effigy 3
( 1 )
T f° (≡ T m°) when Ratel°→s° = Rates°→fifty°
The fact that these 2 phases are in equilibrium does non hateful that molecules are not passing from one phase to the other. The equilibrium is dynamic. Molecules are constantly passing from one phase into the other but the rates of these processes are equal. At equilibrium, the number of molecules leaving and entering the solid phase per unit time are equal. Suppose a solute is dissolved into the liquid. The solute volition not enter the solid phase because the incorporation of solute molecules into the crystal structure of the solid solvent is commonly energetically unfavorable. This keeps impurities (the solute) out of the solid phase as the liquid freezes. The solute remains in the liquid phase just. A molecular view is helpful in agreement what is observed experimentally. To do this, nosotros need to define entropy. Entropy is a measure out of the disorder of a system. College entropy is more matted (e.g., a liquid); lower entropy is less disordered (due east.1000., a solid). Stage changes involve changes in both energy and in entropy. At the freezing point, the kinetic energy of the particles becomes depression enough for inter-particle forces to lock the particles into fixed positions in the solid crystal structure. Nonetheless, if solute particles are present, the entropy of the liquid phase is increased. To attain the freezing point of the solution, the temperature has to be lower than it is in the case of the pure solvent. The system must reach the point where the energy and entropy are low enough for the inter-particle forces to accept control and gear up the sovent particles in position in the crystal structure. Therefore, the temperature at which the solution freezes is lower than that of the pure solvent. The change in the freezing point is proportional to the amount of solute added. This phenomenon is called freezing bespeak depression. Another way to retrieve near freezing point low is to consider the solid surface area available to the solvent molecules. Solute particles are present at the liquid-solid interface, finer decreasing the expanse of the solid accessible to the solvent molecules. This decreases the charge per unit at which liquid-stage solvent molecules tin can stick to the solid. The solute molecules do zilch to the rate at which sufficiently energetic molecules exit the solid phase. The rate of freezing is slowed while the rate of melting is unchanged. As the temperature is lowered, the rate of freezing (sticking) is increased, and the rate of melting is decreased. At some temperature below the freezing point of the pure solvent, the rates of freezing and melting will be equal (freezing point low).
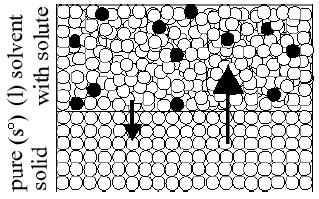
Effigy 4
The modify in the freezing point is defined equally: Δ T f = T f,solution − T f,solvent.Δ T f
( 2 )
molality ≡
≡ = [solute] moles of solute mass of solvent in kilograms
The magnitude of the freezing bespeak change is proportional to the molality of the dissolved solute: Δ T f∝
( 3 )
Δ T f,solution − Δ T f,solvent = Δ T f = − K f [solute] = − K f
The minus sign in the last ii expressions in Eq. three Δ T f,solution − Δ T f,solvent = Δ T f = − Thou f [solute] = − K f Δ T f,solution − Δ T f,solvent = Δ T f = − Thousand f [solute] = − Grand f Δ T f,solution − Δ T f,solvent = Δ T f = − K f [solute] = − G f Δ T f,solution − Δ T f,solvent = Δ T f = − K f [solute] = − K f
( 4 )
Δ T f = − K f
Because the mass of unknown solute is known (measured on the balance) and the number of moles has been calculated (by Eq. 4 Δ T f = − G f
You volition be working with cyclohexane as your solvent.
formula = C6H12 MM = 84.160 g/mol ρ = 0.779 g/mL at 20°C T f = vi.5°C K f = 20.two°C/mol/kg
Procedure
Caution:
Cyclohexane is highly combustible. No flames are permitted in the lab.
Caution:
para-Dichlorobenzene and the organic unknowns are irritants and should not be inhaled or immune to touch the skin.
Caution:
Dispose of all solutions every bit instructed. Nothing may go down the sink.
Caution:
Habiliment your goggles at all times, including trips to the stockroom.
Caution:
Exist conscientious with the thermometer. Report a mercury spill: it must be cleaned upward immediately.
To minimize reagent contagion, never put annihilation into a reagent bottle. Never pipet out of a reagent bottle. Transfer some liquid into a beaker and pipet out of that. Delight use the pipets that are out on the reagent demote. Do not wash them with water. Please return them when you lot are done. The freezing point apparatus is shown in effigy v.
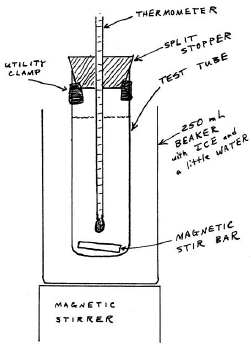
Figure 5 : Freezing-Bespeak Apparatus
one
Carefully conform the height of the thermometer so the entire mercury bulb is immersed in the liquid but so that there is room for the stir bar beneath it.
2
Carefully rotate the carve up stopper so the graduations on the thermometer are visible through the split. Make certain the utility clamp is located and then that you can easily read the thermometer.
3
Prepare an ice-water slurry. Fill up your 250-mL beaker with ice, add plenty water to make full the spaces, and stir. The temperature should be inside a degree or ii of 0°C.
4
Make clean your test tubes and dry thoroughly. The tube must be completely dry earlier cyclohexane is added. Rinse the clean exam tubes with 1-2 mL cyclohexane to remove impurities; dispose rinse liquid properly in the hood. Dry the test tube.
The liquid waste bottle in the fume hood is for minor cyclohexane rinse volumes simply, NOT USED SOLUTION.
Office ane: Determination of K f for Cyclohexane
The freezing betoken must be within the temperature range spanned past your solutions of known solute molality. It is also important that part 2 is done well. Practice part 2 kickoff and repeat information technology again terminal.
A. Pure cyclohexane:
1
Weigh the first empty tube, stir bar included, to 0.01 g (pan balance). It may exist easier to put the test tube in a beaker to have the mass.
2
Pipet in twenty mL of cyclohexane and reweigh to 0.01 grand.
three
Stopper the test tube with the split stopper and adapt the apparatus so that 1) the thermometer does non touch on the side of the test tube, and two) all of the solution in the tube is immersed below the surface of the water ice bathroom.
4
Work with a partner to record the fourth dimension and temperature information. Exercise doing this quickly and accurately. Start timing when the temperature gets but in a higher place 13°C. Record the temperature to the tenths place every bit accurately every bit possible every five seconds. Be prepared to read and tape quickly.
five
Eventually, the modify in temperature will slow enough that readings can be taken every 10 seconds. When the liquid starts to freeze, the temperature change will be very small. Take several information points after this happens (temperature readings are roughly constant). You should have two significant figures in your freezing point temperature.
half dozen
If you need to echo a trial to get better data, warm the test tube and so put it back into the water ice bath. Repeat the experiment enough times so that your information are authentic and reproducible.
B. para-Dichlorobenzene solutions of known concentrations:
Stop. Delight check with your TA if you are to do this section. If yous are instructed to go to part 2, delight utilize the literature value of 1000 f for cyclohexane (20.ii°C/molal).
1
Precisely weigh enough p-dichlorobenzene to make a roughly 0.05 molal solution when added to your 20 mL of cyclohexane.
2
Weigh the solid on the analytical balance. Brand sure that all of the solid is transferred into the test tube and that information technology completely dissolves earlier making any temperature measurements. Do not weigh out the p-dichlorobenzene in advance of using it considering it is a volatile solid (it sublimes quite speedily).
iii
One time your solution is prepared, accept time-temperature data.
4
Accurately weigh plenty p-dichlorobenzene to make a roughly 0.10 molal solution when added to the solution you already accept in your test tube. Dissolve the additional solid in your solution and take fourth dimension-temperature information.
five
Prepare and take fourth dimension-temperature information for an approximately 0.15 molal solution.
6
Determine the freezing bespeak of the pure solvent and of each solution of known molality: Make freezing point (time-temperature) plots — inquire your TA for the number of plots required. a b c
seven
Make up one's mind the molal freezing point depression abiding, K f: a b −Δ T f −Δ T f Δ T f c d Δ T f,solution − Δ T f,solvent = Δ T f = − K f [solute] = − K f due east
When yous are done: Stopper your test tubes. Characterization them with the contents, your name, and your section. Immediately turn them in to the stockroom for disposal. Improper disposal of your organic waste product solutions will event in your immediate ejection from the lab and from the form.
Part two: Molar Mass of an Unknown Organic Solid (para-dihalobenzene)
1
Weigh the second clean, dry out examination tube with a stir bar to 0.01 1000.
two
Pipet in 20 mL cyclohexane and reweigh. Exercise another conclusion of the freezing signal of the pure solvent earlier adding the unknown solute (i.e., take time-temperature measurements for this sample of pure solvent).
3
To determine the freezing point of a solution with a known mass of unknown solute, accurately weigh about 0.37 g of your unknown organic solid on the analytical balance (to 0.0001 g). This mass will produce a sufficient freezing point change while minimizing waste.
4
Completely dissolve the unknown solid in the cyclohexane. Take fourth dimension-temperature data and detect the new freezing point of the solution to two meaning figures.
5
Use One thousand f (to three significant figures) and your freezing point alter (to two meaning figures) to calculate the number of moles and then the molar mass of your unknown solid (2 significant figures).
6
The unknown is a para-dihalobenzene (halo = fluoro, chloro, bromo, or iodo). Report the identity of your unknown based on the molar mass.
When you are done: Stopper your exam tube. Label information technology with the contents, your proper noun, and your department. Immediately plow it in to the stockroom for disposal. Improper disposal of your organic waste solutions will consequence in your firsthand ejection from the lab and from the course.
Reporting Results
Complete your lab summary or write a report (every bit instructed).
Results
- At least two graphs of freezing signal determinations (temperature vs. time) for pure cyclohexane
- At to the lowest degree 1 graph each for ~0.05, ~0.10, ~0.15 molal p-dichlorobenzene
- At to the lowest degree i graph for ~0.37 g unknown / xx mL cyclohexane
- Results table of
T f, Δ T f
, and calculated molalities - Thousand f determination graph (
−Δ T f
versus molality of solute) with calculated K f value. - Report tooth mass and identity of the unknown organic solid
Sample Calculations:
- Thou f determination
- Molar Mass conclusion
Word/Conclusions
- What did you detect out and how?
- Why do impurities lower T f?
- How might this experiment be inverse to improve its accurateness?
Review
kirkhamthavinct54.blogspot.com
Source: https://www.webassign.net/labsgraceperiod/ucscgencheml1/lab_7/manual.html
0 Response to "what would happen to the molality of cyclohexane if some of it evaporated"
Enregistrer un commentaire